Background
Ethylene oxide, or EtO, is a manufactured and naturally occurring chemical that has numerous important commercial uses. EtO is used to produce antifreeze and polyester, as an insecticide on stored agricultural products, and to sterilize food, cosmetics, and medical equipment and supplies. However, like many other useful chemicals EtO can be hazardous to human health at elevated levels of exposure over long periods of time.
Research into EtO toxicity led to implementation of workplace and environmental regulations in the 1980s by the Occupational Safety and Health Administration (OSHA)1 and U.S. Environmental Protection Agency (USEPA)23. Following these actions, newly regulated parties integrated the federal rules into their routine operations and state agencies did as well.
Meanwhile, new studies of associations between EtO exposure among people at work and different types of cancers began to appear in the literature. A series of publications from a longitudinal cohort study of sterilization facility workers conducted by the National Institute of Occupational Safety and Health (NIOSH) received particular attention.4567 Effects of those studies and others rippled through programmatic analyses and regulatory processes at USEPA circa 2006 - 2016.8 Those Agency efforts preceded more recent actions by USEPA, state and local authorities, and communities that have returned EtO to the spotlight.9
The renewed attention has created diverse but complementary needs for reliable information on how to manage EtO effectively.
Producers and users of EtO need to understand requirements for complying with applicable regulations, possible changes to requirements for its handling and disposal, the latest evidence for EtO toxicity, and the performance of their current workplace and environmental controls.
Regulators need to understand sources and pathways of total human exposure to EtO and efficacy of control options so they can identify policies that ensure workers and communities are protected from meaningful risks to health.
The public needs reliable information that puts potential health risks of EtO in context and supports informed perspectives and decisions.
The aim of this post is to introduce those groups to information that is foundational to sound understanding and management of environmental and occupational health concerns of EtO. We first address several intrinsic characteristics of EtO and then branch into questions related to workplace and community settings.
1. Chemical Structure and State
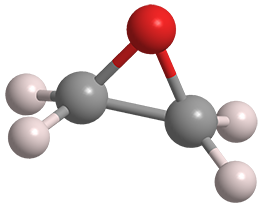
Ethylene oxide is composed of two carbon atoms, four hydrogen atoms, and one oxygen atom. Given that structure, EtO has the chemical formula C2H4O and molecular weight of 44 grams per mole, placing it in a group of comparatively small molecules within the universe of chemicals.
EtO is distinguished from other substances with the same chemical formula by the three-sided ring of carbon (grey) and oxygen (red) atoms shown in the figure below. The nature of the ring allows EtO to easily react with other substances and contributes to its ability to inactivate bacteria, fungi, and viruses.
As with many other relatively small molecules primarily composed of carbon and hydrogen, EtO is a gas at ordinary environmental temperatures and pressures. EtO is colorless and odorless except at very high concentrations where it has a sweet smell. Not surprisingly then, EtO is primarily released to the environment as a gas.
Largely because of the oxygen atom in its structure, EtO also mixes easily with water. Its water solubility has some practical applications. For instance, a category of air pollution control systems commonly known as scrubbers work by spraying water droplets onto an EtO-containing gas stream; the droplets intercept molecules of EtO and carry them down to a reservoir at the base of the scrubber. In the atmosphere, rain will have a similar effect and scavenge EtO from the atmosphere.
2. Sources
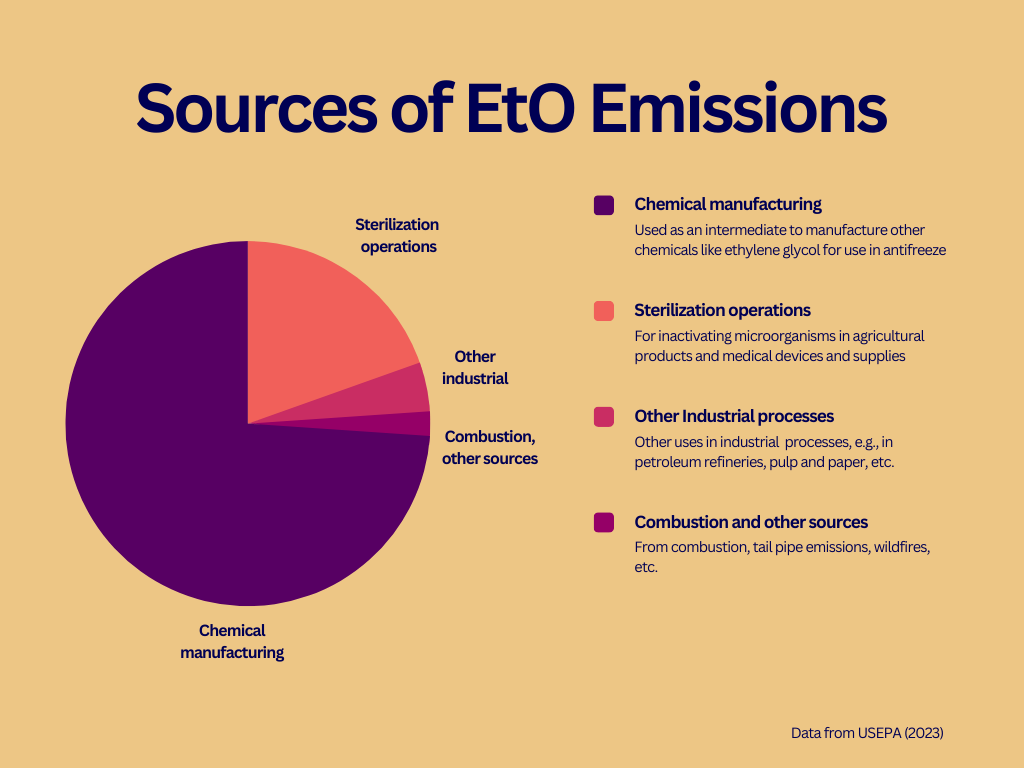
EtO is produced by both industrial and natural sources. Chemical manufacturers in the United States produce approximately 6,400 million pounds of EtO annually. The majority of EtO produced in the US is used to manufacture other chemicals, such as ethylene glycol. Much of the remaining EtO is used to inactivate microorganisms in agricultural products and medical devices and supplies, i.e., to sterilize those products.
According to EPA, commercial sources released 135,088 pounds of EtO to the atmosphere in 2020,10 which represents less than one-tenth of one percent of the EtO produced each year. Chemical manufacturers are estimated to account for nearly three-quarters of EtO emissions, while facilities that sterilize medical equipment and supplies or agricultural products released about one-fifth of the total emissions.
In addition to releases from commercial operations, EtO is reported to be a byproduct of hydrocarbon fuel combustion.11 USEPA states that its understanding of emissions from combustion is insufficient and plans to test for EtO in tailpipe emissions from motor vehicles,12 although a timeline for that research has not been released as of this post.
EtO is also produced within the human body as a result of normal metabolism. This process is described below in the context of exposure.
3. Hazards
The principal hazards of EtO are its explosivity, flammability and toxicity.
EtO is explosive and flammable at concentrations in air above 3%. Therefore, industrial processes that use concentrated EtO are designed and operated to avoid conditions that present a risk of ignition. Concentrated EtO is contained in sealed piping and vessels rather than used in open systems. Only tools made of non-sparking metals are allowed in most areas where concentrated EtO could be present. Similarly, electronic devices like cell phones are generally prohibited from those areas. While only a few industrial accidents involving ethylene oxide have been reported, at least three people have died and more have been injured in explosions at EtO production or sterilization facilities which indicates the importance of safe management of this chemical.
Largely because of its mutagenicity and evidence for human carcinogenicity at high doses, multiple government agencies and other organizations identify EtO as a carcinogen. For instance, the U.S. Department of Health and Human Services considers EtO to be a known human carcinogen. Similarly, the International Agency for Research on Cancer designates ethylene oxide as a Group 1 human carcinogen. USEPA concluded that EtO is carcinogenic to humans based on 1) epidemiological evidence of lymphohematopoietic cancers and breast cancers in EtO-exposed workers; 2) evidence of carcinogenicity in laboratory animals; 3) evidence that EtO is genotoxic and supports a mutagenic mode of action; and 4) evidence that EtO can lead to chromosome damage in humans.
4. Exposures
The major routes of human exposure to EtO are thought to be: (1) metabolism and (2) inhalation. While EtO has been reported in certain foods, intake via ingestion is likely negligible compared to inhalation and metabolism.
EtO exposure can be assessed directly by measuring amounts of compounds formed when EtO binds with DNA or hemoglobin. These compounds are called adducts. The primary adducts specific to EtO are N7-HEG [full name N7-(2-hydroxyethyl)guanine] (N7-HEG) and HEV [full name 2-hydroxyethylvaline]. As with other biomarkers, these adducts reflect intake of EtO from all exposure routes and pathways.
The US Centers for Disease Control and Prevention (CDC) routinely measures EtO adducts in the general population.13 One take away point from CDC’s surveys is that essentially all people in the United States have measurable levels of HEV adducts. In other words, we are all exposed to an observable level of EtO.
The ways in which EtO exposure can occur and their relative magnitude are summarized next.
Endogenous production
Humans produce EtO inside their bodies. EtO produced internally is termed endogenous, which means having an internal cause or origin. Endogenous EtO is a byproduct of metabolism of ethylene. This EtO exposure is unavoidable because ethylene is produced in the body as well. The amount of EtO formed inside the body is estimated to be equivalent to inhaling approximately 2 to 3 parts per billion by volume of airborne EtO.14 That concentration is about 10-fold higher than average EtO levels in outdoor air.
Ambient air
Research shows that EtO is present in outdoor air across the United States. For example, testing by USEPA at 51 locations around the country in 2021 showed an average EtO concentration of approximately 0.2 parts per billion. Here is a link to USEPA monitoring data from 18 sites in nine states during 2018 - 2019 that produced similar findings. Because of its ubiquity, inhalation of EtO in ambient air likely occurs continuously for most people.
Smoking
Smokers have substantially elevated EtO exposure relative to non-smokers. HEV adducts demonstrate the difference quite clearly; average concentrations in active smokers are approximately 200 picomoles of HEV per gram (pmol/g) of hemoglobin compared to only about 20 pmol/g for non-smokers.15 These findings indicate that a fraction of the population voluntarily spends money on a product that causes them to be exposed to elevated amounts of EtO, and of course other toxic substances.
Workplaces
Some workers experience elevated exposure to EtO compared to the general population. This group includes people who work in areas where EtO is manufactured or used to sterilize products. The amount of published data on current workplace exposure concentrations is limited, but earlier research indicates that levels have trended downward over time and are generally below the OSHA standard of 1000 parts per billion over an 8-hour period.16
Facility Emissions
Facilities that manufacture EtO or sterilize with EtO have permits issued by states that allow them to release limited amounts of EtO to outdoor air. Depending on their magnitude, those emissions have the potential to create elevated EtO concentrations in outdoor air of locations nearby.
USEPA regularly evaluates possible air quality impacts of EtO emissions from facilities. What is perhaps the most well-developed of those programs was originally known as the National Air Toxics Assessment and is now called AirToxScreen. As part of that program, USEPA estimates annual average EtO concentrations for each census tract in the US. The Agency’s estimates are made with a widely accepted air dispersion model known as AERMOD and EtO emissions reported by each facility.
EPA's most recent analysis is based on emission rates for calendar year 2018. The census tracts with the top 25 highest modeled annual average EtO concentrations based on 2018 emissions are tabulated below. The EtO concentrations range from 0.018 parts per billion by volume [ppb(v)] to 0.030 ppb(v). One observation to make from the table is that all of the modeled concentrations are less than 0.2 ppb(v), the average EtO concentration in outdoor air that USEPA measured at 51 locations across the country in 2021 and a smaller set of locations in 2018 - 2019; see section on Ambient Air. Said another way, these modeling results indicate that census tract-average EtO concentrations potentially attributable to emissions from commercial facilities are likely lower than ambient or background levels of EtO. Here is a link to the full set of AirToxScreen results from USEPA.
5. Health risks
A widely applied science policy convention in public health is that a linear relationship exists between the cumulative dose of a chemical carcinogen and the probability of a cancerous tumor forming from that exposure. Cumulative dose is the total amount of a chemical inhaled or ingested over a certain time period, e.g., the amount of EtO inhaled over a lifetime. Generally speaking, the higher the long-term average exposure concentration holding duration constant and the longer the duration of exposure holding exposure concentration constant, the larger the cumulative dose.
Based on the exposure concentration and duration information presented in #4:
The cumulative dose from internally generated EtO appears to be substantially greater than that from sources outside the body for the general population.
Cumulative doses of EtO for people exposed at work or through smoking can be substantially greater than from inhalation of EtO in ambient air.
Extensive air modeling by USEPA indicates that cumulative doses of EtO for most people as a result of emissions from commercial sources are likely generally lower than doses from inhalation of background levels in outdoor air.
Summary of Key Points
Ethylene oxide is a naturally occurring and manufactured chemical commonly present in outdoor air and used in several important commercial processes.
The principal commercial sources of EtO emissions are reported to be chemical production and sterilization of medical devices. Other emission sources, such as combustion of fossil fuels or biomass, are less well characterized but the subject of current research.
Important hazardous properties of EtO are its flammability and explosivity at high concentrations, and toxicity. Numerous government and non-government organizations classify EtO as a carcinogen.
Human exposure to EtO occurs through several different pathways, including but not limited to:
production within the human body
tobacco smoking
workplaces such as certain chemical manufacturing and sterilization facilities
background levels in outdoor air
emissions from commercial operations
For a typical person in the US, the risk of cancer related to EtO appears to be driven largely by background levels in ambient air and endogenous production of EtO.
Cancer risks can be higher, perhaps substantially so, for people who smoke or work in chemical manufacturing or commercial sterilization operations.
Cancer risks associated with emissions from commercial sources and based on USEPA air modeling appear to be low in general compared to other sources and exposure pathways.
In the next few posts, we will take a deeper look at the hazards and the health risks of EtO, with some discussion of USEPA modeling and how to contextualize risk from EtO relative to other naturally occurring chemicals. We will also share a dashboard that we have been developing to allow users to understand risk from EtO in their communities.
OSHA. OSHA 1910.1047. Ethylene oxide. Accessed January 10, 2023. https://www.osha.gov/laws-regs/regulations/standardnumber/1910/1910.1047
USEPA. Health Assessment Document for Ethylene Oxide. U.S. Environmental Protection Agency; 1985:1-302. https://cfpub.epa.gov/ncea/iris_drafts/recordisplay.cfm?deid=41115
USEPA. 40 CFR Part 63 Subpart O. Code of Federal Regulations. Accessed January 10, 2023. https://www.ecfr.gov/current/title-40/chapter-I/subchapter-C/part-63/subpart-O
Steenland K, Stayner L. An epidemiological study of workers potentially exposed to ethylene oxide. Br J Ind Med. 1993;50(12):1125-1126. doi:10.1136/oem.50.12.1125
Steenland K, Whelan E, Deddens J, Stayner L, Ward E. Ethylene oxide and breast cancer incidence in a cohort study of 7576 women (United States). Cancer Causes Control. 2003;14(6):531-539. doi:10.1023/a:1024891529592
Stayner L, Steenland K, Greife A, et al. Exposure-response analysis of cancer mortality in a cohort of workers exposed to ethylene oxide. Am J Epidemiol. 1993;138(10):787-798. doi:10.1093/oxfordjournals.aje.a116782
Steenland K, Stayner L, Deddens J. Mortality analyses in a cohort of 18 235 ethylene oxide exposed workers: follow up extended from 1987 to 1998. Occup Environ Med. 2004;61(1):2-7. https://www.ncbi.nlm.nih.gov/pubmed/14691266
USEPA. History/chronology. Ethylene oxide CASRN 75-21-8. Published January 30, 2017. Accessed December 10, 2022. https://cfpub.epa.gov/ncea/iris_drafts/recordisplay.cfm?deid=329730
USEPA. What EPA is Doing to Address Ethylene Oxide. Hazardous Air Pollutants: Ethylene Oxide. Published December 22, 2022. Accessed January 5, 2023. https://www.epa.gov/hazardous-air-pollutants-ethylene-oxide/what-epa-doing-address-ethylene-oxide-and-learn-more-about#:~:text=After%20conducting%20a%20%E2%80%9Crisk%20and,by%20multiple%20air%20toxics%20rules.
USEPA. Find TRI Data. Toxics Release Inventory Program. Published January 25, 2023. Accessed January 25, 2023. https://www.epa.gov/toxics-release-inventory-tri-program/find-understand-and-use-tri
ATSDR. Toxicological Profile for Ethylene Oxide. Agency for Toxic Substances and Disease Registry; 2022:106. Accessed December 5, 2022. https://www.atsdr.cdc.gov/ToxProfiles/tp137-c5.pdf
USEPA. What EPA is Doing to Address Ethylene Oxide. Hazardous Air Pollutants: Ethylene Oxide. Published December 22, 2022. Accessed January 5, 2023.
CDC. Biomonitoring Data Tables for Environmental Chemicals. CDC Exposure Report. Published March 4, 2022. Accessed December 22, 2022. https://www.cdc.gov/exposurereport/data_tables.html
Kirman C, Li A, Sheehan P, Bus J, Lewis R, Hays S. Ethylene oxide review: characterization of total exposure via endogenous and exogenous pathways and their implications to risk assessment and risk management. J Toxicol Environ Health B Crit Rev. 2021;24(1):1-29. doi:10.1080/10937404.2020.1852988
CDC. Biomonitoring Data Tables for Environmental Chemicals. CDC Exposure Report. Published March 4, 2022. Accessed December 22, 2022. https://www.cdc.gov/exposurereport/data_tables.html
LaMontagne AD, Oakes JM, Lopez Turley RN. Long-term ethylene oxide exposure trends in US hospitals: relationship with OSHA regulatory and enforcement actions. Am J Public Health. 2004 Sep;94(9):1614-9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1448503/